The 3rd International Electronic Conference on Antibiotics
Rise of Antibiotic Resistance: Mechanisms Involved and Solutions to Tackle it
Part of the International Electronic Conference on Antibiotics series
1–15 December 2023
Antibiotic, antimicrobial molecules, Drug Resistance, Repurposing, mode of action, Antibiotic Stewardship
- Go to the Sessions
-
- S1. Epidemiology, Prevalence and Mechanisms of Microbial Resistance
- S2. Making Old Antibiotics Great Again: How to Reverse Resistance to Antibiotics
- S3. New Antimicrobial Approaches, Targets and Mechanisms of Action
- S4. Social Challenges and Political Strategies to Prevent Antibiotic Resistance
- S5. Antimicrobial Testing and Rapid Diagnostics
- S6. Impact of Inter-relationship between Viral and Bacterial Infections
- S7. Poster Session
- Event Details
ECA 2023 is closed. Thank you for your participation.
The accepted papers will be published as one dedicated volume in the MDPI Medical Sciences Forum journal (ISSN: 2673-9992). Publication of proceedings paper is free of charge.
Certificates of participation are available HERE.
The open access journal Antibiotics published a dedicated conference Special Issue: Rise of Antibiotic Resistance: Mechanisms Involved and Solutions to Tackle It. The conference participants are encouraged to submit a full paper to the dedicated Special Issue and will receive a 20% discount.
Live Session Program
ECA 2023 | Live Session I
Date: 1 December 2023
|
| Time in CET | Speaker | Title |
| 14:00-14:10 | Prof. Dr. Manuel Simões University of Porto, Portugal |
Opening Speech of ECA 2023. |
| 14:10-14:40 | Prof. Dr. Manuel Simões University of Porto, Portugal |
Plant chemicals in biofilm control. |
| 14:40-15:10 | Prof. Dr. Jordi Vila Hospital Clinic, Spain |
Strategies to design new antibacterial agents. |
| 15:10-15:20 | Discussion and Q&A | |
ECA 2023 | Live Session II
Date: 8 December 2023
|
| Time in CET | Speaker | Title |
| 10:00-10:30 | Prof. Dr. Rafael Cantón Ramón y Cajal's University Hospital, Spain |
Epidemiology of antimicrobial resistance and emerging threats. |
| 10:30-11:00 | Prof. Dr. Efstathios Giaouris University of the Aegean, Greece |
Prevalence of important foodborne pathogenic bacteria in retail chilled raw chicken meat and antibiotic resistance of Campylobacter spp. isolates. |
| 11:00-11:10 | Discussion and Q&A |
|
ECA 2023 | Live Session III
Date: 15 December 2023
|
| Time in CET | Speaker | Title |
| 14:00-14:30 | Prof. Dr. Kim Lewis Northeastern University, USA |
Keynote Speech: The quest for new antibiotics. |
| 14:30-15:00 | Prof. Dr. Christian Giske Karolinska Insititute, Sweden |
Novel gram-negative antimicrobials. |
| 15:00-15:30 | Dr. Jean-Michel Bolla Aix-Marseille Universite, France |
How to hijack antibiotic resistance: from nature to chemistry. |
| 15:30-16:00 | Prof. Dr. Octávio Franco Brasília Catholic University, Brazil |
In Silico Strategies for Developing Novel Antimicrobial Peptides with Industrial Potential. |
| 16:00-16:10 | Discussion and Q&A | |
Invited Speaker

Strategies to design new antibacterial agents.
Dr. Vila is currently the Head of the Department of Clinical Microbiology of the Hospital Clinic in Barcelona, Full Professor of the School of Medicine, University of Barcelona, and Research Professor in the Institute for Global Health (ISGlobal) of Barcelona, Spain. His present main field of interest is in the investigation of the molecular bases of antimicrobial resistance and the development of new drugs against bacteria and molecular tools for rapid diagnosis of infectious diseases. Dr. Vila was the Programme Director of the Congress of the European Society of Clinical Microbiology (ESCMID) from 2009 to 2014 and he has been a member of the ICAAC Scientific Program Committee (USA) for 4 years. He received an Award of the National Plan against Antibiotic Resistance (PRAN) 2018 for the Micro-combat card game, presented by the Barcelona Global Health Institute Foundation (ISGlobal) in the category of better communication and public awareness initiative on the antibiotic resistance. He is also leading the Initiative of Antimicrobial Resistance in ISGlobal. He has published 476 articles in peer-reviewed journals, 44 book chapters and directed 25 doctoral theses (number of citations, 29.536 and H-index of 84, Google Scholar). He has patented three molecules.

Antimicrobial Discovery Center, Department of Biology, Northeastern University, USA
The Quest For New Antibiotics.
Kim Lewis is a University Distinguished Professor and Director, Antimicrobial Discovery Center at Northeastern University in Boston. He is a Fellow of the American Society of Microbiology, and a Fellow of the American Association for the Advancement of Science. He is a Highly Cited Researcher (Clarivate Analytics) and an Expertscape World Expert in Microbial Drug Resistance (top 0.1% of scholars in the field). He obtained his Ph.D. in Biochemistry from Moscow University in 1980, and has been on the Faculty of MIT, University of Maryland, and Tufts University prior to coming to Northeastern. Dr. Lewis has authored over 100 papers and is an inventor on several patents. His notable findings include the development of general methods to grow previously uncultured bacteria that make up >99% of biodiversity on the planet, the discovery of the culprit of recalcitrant biofilm infections, drug-tolerant persister cells; and several novel antibiotics, including teixobactin and darobactin. Dr. Lewis has served as a panelist and contributor to reports on antimicrobial resistance (AMR) by National Academies Institute of Medicine, the Pew Charitable Trust, and the European Academies of Science. Dr. Lewis is a member of Faculty 1000, a world-wide panel of experts evaluating research advancements. He is a recipient of the MIT C.E. Reed Faculty Initiative Award, the NIH Director’s Transformative Award, and the American Society for Microbilogy Applied Biology and Biotechnology Research Award. Apart from his work in Academia, Dr. Lewis has served as a consultant to the Pharmaceutical Industry, The Biotech, and is a co-founder of NovoBiotic Pharmaceuticals, Arietis Pharma, Holobiome, Flightpath and Odyssey Therapeutics.

Ramón y Cajal's University Hospital, Spain
Epidemiology of antimicrobial resistance and emerging threats
Rafael Cantón is a Clinical Microbiology specialist, Head of the Clinical Microbiology Department at the University Hospital Ramón y Cajal in Madrid (Spain) and associated Professor of Clinical Microbiology at Complutense University in Madrid. His research activity is developed within the Spanish Network CIBER in Infectious Diseases (CIBERINFEC, https://www.ciberinfec.es/en/groups/research-group?id=27645) and Institute Ramón y Cajal for Health Research (IRYCIS, https://www.irycis.org/en/research/research-groups/6/biology-and-evolution-of-microorganisms) in which he coordinates the Microbiology, Immunology and Infection Area. This activity is focused on antimicrobial resistance mechanisms, including extended-spectrum beta-lactamases and carbapenemases, antimicrobial susceptibility testing with new technology, respiratory tract infections (mainly in bronchiectasis and cystic fibrosis), and interplay of clonal epidemiology and resistance. He is currently the clinical data coordinator of the European Committee for Antimicrobial Susceptibility Testing (EUCAST) (former president 2012 - 2016). He has been President of the Spanish Society of Infectious Diseases and Clinical Microbiology (SEIMC) (2015-2017). Since 2018 he is in the Advisory Board of the Joint Programing Initiative on Antimicrobial Resistance (JPIAMR). He has several publications in peer-review journals, book chapters and frequently participate on international meeting and congresses. He is associated editor of Diagnostic Microbiology and Infectious Diseases journal and member of the editorial board of Microbial Drug Resistant, Drug Resistance Updates, Enfermedades Infecciosas y Microbiología Clinica and Revista Española de Quimioterapia journals. He has been editor of the Clinical Microbiology Procedures of the SEIMC (https://seimc.org/).

Karolinska Insititute, Solna, Sweden
Novel Gram-negative antimicrobials

Proteomics and Biochemical Analysis Center, Brasília Catholic University, Brazil,
S-Inova Biotech, Pós-Graduação em Biotecnologia, Universidade Católica Dom Bosco, Brazil
In silico strategies for developing novel antimicrobial peptides with industrial potential

Faculté de pharmacie, Aix-Marseille Universite, France

Department of Food Science and Nutrition, Faculty of the Environment, University of the Aegean, Greece
Prevalence of important foodborne pathogenic bacteria in retail chilled raw chicken meat and antibiotic resistance of Campylobacter spp. isolates
List of accepted submissions (77)
| Id | Title | Authors | Presentation Video | Poster PDF | |||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| sciforum-080250 | Novel natural and synthetic anticandidal therapeutic peptides to combat drug resistant infections | N/A |

|
Show Abstract |
|||||||||||||||||||||||||||||||||||||
|
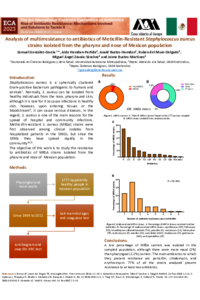
Candida species are considered as common flora of the healthy human mycobiome and occur on skin, mucosal surfaces of gastrointestinal and genitourinary tracks. Pathogenic Candida spp., reported to cause skin, vaginal and oral infections as well as systemic infection in immunocompromised humans. Extensive use of antifungal agents has increased the drug resistance among pathogenic strains of Candida. The biofilm formation ability of Candida strains also adds on resistance to antifungal drugs. This increased resistant desperately requires alternative antifungal agents to overcome this problem. To this effect, recently naturally occurring AMPs and synthetically modified peptides, are effectively being used as promising antifungal agents. Short peptides display better permeability to cross the yeast membrane, thus short antifungal peptide were designed using sequences from APD database. Antifungal potential of designed peptides was predicted in terms of protein binding potential, amphipathicity, hydrophobicity, hydrophilicity and net charge. In vitro synthetic peptides (SK08 and pep2) showed anticandidal activity against various test strains. Natural peptides are potent antifungal agents, considering these facts, we focused on screening potent antifungal peptides from Bacillus sp. strain A52 and Bacillus sp. strain SVDS-15 that were found to inhibit various pathogenic Candida strains. However, both of these lipopeptides exhibited hemolytic activity. Interestingly, the lipopeptides did not show any phytotoxic effect in seed germination experiments. Further, emulgel formulations were developed containing cetomacrogol 1000 and lipopeptides (A52 and SVDS-15) which showed in vitro, antimicrobial activity. No irritation effects were observed with formulation during animal studies using BALB/c mice indicating their potential for external therapeutic applications.
|
|||||||||||||||||||||||||||||||||||||||||
| sciforum-080487 | METALLO-β-LACTAMASE PRODUCING GRAM-NEGATIVE BACTERIA ISOLATED FROM CHICKEN MEAT IN BHARATPUR, CHITWAN | , , , | N/A | N/A |
Show Abstract |
||||||||||||||||||||||||||||||||||||
|
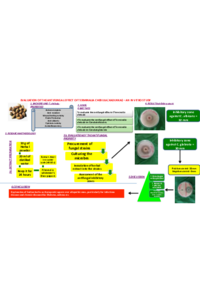
A rising threat of the rapid spread of acquired metallo-beta-lactamases (MBLs) among major Gram-negative pathogens is a matter of public health concern worldwide. The current study was undertaken to investigate the prevalence of Metallo-beta-lactamase producing gram-negative bacteria isolated from chicken meat in Bharatpur. The study was conducted for the period of 3 months from April 2023 to June 2023 in the Microbiology laboratory of Balkumari College, Bharatpur, Chitwan. Among the 40 collected sample, 12 (30%) isolates were E. coli, 10(25%) Citrobacter ,8(20%) Salmonella spp., 4(10%) Proteus spp., 4(10%) Shigella spp. and 2(5%) Klebsiella spp. Out of 12 isolates of E. coli species 10 (83.34%) isolates were found Multidrug resistance and 8 isolates were MBL positive. Out of 10 isolates of Citrobacter species, 7(70%) were found Multidrug resistance and 9 isolates were MBL positive. Similarly, out of 8 isolates of Salmonella species, 6(75%) were found to be Multidrug resistance and 5 isolates were MBL positive. Among the 4 isolates of Proteus species, 2(50%) were found to be Multidrug resistance and 3 were MBL positive. Out of 4 isolates of Shigella species, 3(75%) isolates were found to be Multidrug resistance and 3 isolates were MBL positive. Likewise, out of 2 isolates of Klebsiella species, 1 was found to be Multidrug resistance and both isolates were MBL positive. |
|||||||||||||||||||||||||||||||||||||||||
| sciforum-080668 | Optimization of Polyvinyl Alcohol and Gum Tragacanth Membrane with Ciprofloxacin-loaded Gold Nanoparticles for Wound Healing Applications | , , , | N/A | N/A |
Show Abstract |
||||||||||||||||||||||||||||||||||||
|
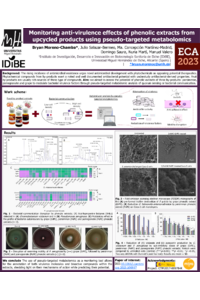
Bandages, Gauzes and Alcoholic pads have widely been used for wound healing. However due to few limitations hydrophilic membranes loaded with nanoparticles have been explored rarely as a wound healing material. This study aimed to synthesize and characterize Polyvinyl Alcohol (PVA) and Gum Tragacanth(GT) membrane having different concentrations of gold nanoparticles(AuNPs) loaded with Ciprofloxacin as bacterial infection is one of the major problem for wound healing. Gold and CIP-loaded nanoparticles were successfully prepared by Turkevich method and confirmed by Fourier-transform infrared spectroscopy. To analyze the hydrophilicity of membrane swelling test as well as contact angle analysis were performed. In summary, this study demonstrated that hydrophilic membrane of PVA/GUM T. having 500ul concentration of CIP-loaded AuNPs can be used as wound healing materials without affecting their biocompatibility. |
|||||||||||||||||||||||||||||||||||||||||
| sciforum-082758 | Highest priority critically important antimicrobial-resistant Escherichia coli and Salmonella spp. isolated from pork and chicken meat from Argentina | , , , , , , , , |

|

|
Show Abstract |
||||||||||||||||||||||||||||||||||||
|
Between June and September 2023, a total of 80 meat samples (48 and 32 from pork and |
|||||||||||||||||||||||||||||||||||||||||
| sciforum-082808 | "Antibiotics in a Changing World: Resistance, Ecology, and Public Health Conundrums" | , , | N/A | N/A |
Show Abstract |
||||||||||||||||||||||||||||||||||||
|
The world of antibiotics is undergoing a significant transformation, driven by the dual challenges of antimicrobial resistance and ecological ramifications associated with their use. This thesis provides a comprehensive examination of the intricate relationship between antibiotics, public health, and the environment, shedding light on the multifaceted problems that this interplay creates. The research scrutinizes the global crisis of antibiotic resistance, investigating its origins, underlying mechanisms, and its alarming proliferation. Antibiotic-resistant bacteria pose a serious threat to the effectiveness of these crucial drugs in the fight against infectious diseases. This necessitates a reevaluation of antibiotic use and the development of innovative strategies to curb resistance also focusing on the ecological consequences of antibiotic utilization. While antibiotics are designed to target specific pathogens, their impact extends to the broader microbial communities they encounter. The research delves into the effects of antibiotics on ecosystem balance, with a particular emphasis on their contribution to the growing problem of environmental antibiotic resistance. This thesis offers a holistic perspective on the evolving landscape of antibiotics through a comprehensive analysis of these interconnected aspects. It emphasizes the urgent need for a global response to address resistance, advocates for a balanced approach to antibiotic usage, and underscores the pivotal role of public health strategies in managing these intricate challenges |
|||||||||||||||||||||||||||||||||||||||||
Welcome from the Chairs

Antibiotics have revolutionized modern medicine and established unprecedented patterns of public and animal health. However, their misuse has exposed bacteria to selective pressure, leading to the development of critical and therapeutically untreatable bacterial resistance. Our current arsenal of effective antibiotics accompanied by the lack of alternatives is prompting the beginning of the “post-antibiotic era”, which threatens all the achievements of modern medicine. The worldwide spread of multidrug-resistant bacteria is increasingly drawing global surveillance from authorities and the media due to its potentially catastrophic implications. The pharmaceutical industry’s limited interest in the discovery of new antibiotics or developing new therapeutic strategies is a critical issue, despite considering the impact such new developments may have on investment returns. Bacteria have demonstrated a rapid ability to develop resistance mechanisms to every new antibiotic. Considerable technological advances, particularly these omics, have allowed for the thorough investigations into and characterization of the mode of action and resistance of antibiotics. Considering the rate of demand, such advances seem to be insufficient in discovering novel antibiotics. Specific and targeted actions are needed to ensure antibiotic sustainability and halt the advance of the “post-antibiotic era”. While the discovery of innovative therapeutic solutions and the comprehension of their action mechanism is fundamental, patient access requirements need to be properly integrated. SARS-COV-2 highlighted the existing interactions between viral and bacterial/fungal infections. However, both the COVID-19 pandemic and Ukrainian/Russian conflict causing a shortage in amoxicillin have presented an unfortunate realization of what the global health situation may become once all commercial antibiotics fail to treat bacterial infections.
Epidemiology, Prevalence and Mechanisms of Microbial Resistance.
Making Old Antibiotics Great Again: How to Reverse Resistance to Antibiotics.
New Antimicrobial Approaches, Targets and Mechanisms of Action.
Social Challenges and Political Strategies to Prevent Antibiotic Resistance.
Antimicrobial Testing and Rapid Diagnostics.
Impact of Inter-relationship between Viral and Bacterial Infections.
Kind regards,
Event Chairs

Department of Chemical Engineering, Faculty of Engineering, University of Porto, Portugal
Manuel Simões has a PhD in Chemical and Biological Engineering and is currently Associate Professor with Habilitation at the Faculty of Engineering of the University of Porto, where he is Pro-Director. He has more than 180 papers published in journals indexed in JCR (h-index = 48), 4 books (1 as author and 3 as editor) and more than 40 chapters in books. His research focusses on biofilms and antimicrobial action and resistance. He has been highlighted as a highly cited author in 2020 and 2021 by Clarivate Analytics. SCOPUS ID: 55608338000; Orcid ID: 0000-0002-3355-4398.

CNRS, Aix-Marseille University, Centrale Marseille, iSm2, Marseille, France
Dr. Marc Maresca is currently a researcher at Aix-Marseille Université. He received his Ph.D. in Biochemistry from the Université Paul Cézanne (France, 2003) working on food contaminants named mycotoxins. In 2003, he moved to England to work on enteropathogenic E coli in Brendan Keny’s lab. Then, he moved back to France to continue his work on mycotoxins and their effects on human health. He is currently working at Aix-Marseille Université and his research aims to identify and develop new molecules—natural, synthetic, or bio-inspired—with antimicrobial properties. He focuses on antimicrobial peptides and their mimics as well as on plant derivatives; in addition to their antimicrobial effects, they may possess additional activities such as anti-inflammatory and anti-tumorigenic effects.
Event Committee

School of Chemistry and Molecular Bioscience, University of Wollongong, NSW 2522, Australia
Prof. Dr. Nick Dixon holds a PhD in Biochemistry from the University of Queensland (1978). He was a Research Fellow with Prof Alan Sargeson (Research School of Chemistry, ANU) before being awarded C.J. Martin and Fulbright Fellowships to study with Nobel laureate Prof Arthur Kornberg at Stanford University, U.S.A. He returned to the John Curtin School of Medical Research at ANU as a Queen Elizabeth II Fellow in 1983, and was appointed as Fellow in Biological Chemistry at the Research School of Chemistry, ANU in 1986. He was subsequently promoted to Senior Fellow and Professor, before leaving ANU in 2006 to take up his present position as Professor of Biological Chemistry at the University of Wollongong. He was awarded an Australian Research Council Australian Professorial Fellowship in 2008, and established UOW’s Centre for Medical and Molecular Bioscience in 2010. He is currently an academic leader of Molecular Horizons, a new facility for molecular visualization which opened in 2019.

Laboratoire des Multimatériaux et Interfaces, Villeurbanne, France
Dr. Anthony W. Coleman studied at Huddersfield New College and qualified by competitive examination for admission to the University of Oxford. He was affiliated to Jesus College from 1972-76 obtaining an Honours degree in Chemistry. Publications 260 in Science Citation Index, 150 Invited and Plenary Talks and 22 Patents (3 licenced).

Jean-Marc SABATIER is a Director of research at the French CNRS, with PhD and HDR degrees in Biochemistry and Microbiology. He has headed several academic research teams (CNRS, INSERM and University), as well as a combined academic–industry research laboratory devoted to the engineering of therapeutic peptides (ERT62, Marseilles, France). He was also a Director of Research for several French private companies as well as a Canadian public company. He acts as a consultant for top pharmaceutical and cosmetic companies. Dr. Sabatier works in the field of animal toxins and microbes. He so far contributed to several books in toxicology and virology, and more than 180 scientific articles, 180 communications, and 55 patents in both biology and chemistry. He acts as the Editor-in-Chief of the journals Coronaviruses, Infectious Disorders – Drug Targets, and Venoms and Toxins. He is a member of 66 Editorial Boards of scientific journals, such as Peptides, Molecules, Antibiotics, Frontiers in Pharmacology, and the Journal of Biological Chemistry. He also reviewed articles submitted for publication in >100 international journals and acts as an expert for numerous national and international institutions. He won the ‘Citizen of the Year Award’ from The Nouvel Economiste (1994) for his work on antivirals. He is a member of a dozen scientific societies, such as the ‘American Peptide Society’ (charter member), ‘European Peptide Society’, ‘American Society for Microbiology’, ‘Biochemical Society’ and ‘New-York Academy of Sciences’.

Dr. Hiroshi Hamamoto studied at the Department of Pharmaceutical Sciences, Kyushu University (1994-1998), and the Grad. Sch. Of Pharm. Sci., Kyushu University (1998-2000). He belonged to the Lab. of Microbiology and studied disinfectant-resistant strains of Staphylococcus aureus. He moved to the Grad. Sch. of Pharm. Sci. at the University of Tokyo for his doctoral studies (2000-2002), during which he established a system to evaluate the quantitative therapeutic effects of antibiotics using a silkworm-based infection system. After graduation, he worked as an assistant professor at the University of Tokyo, where he researched the analysis of pharmacokinetics in silkworm (2003-2005) and the screening of the therapeutically effective antibiotics from soil bacteria culture supernatants using a silkworm bacterial infection model. From 2005 to 2008, He successfully identified a novel antibiotic, Lysocin E, by discovering antibiotics through the use of a silkworm infection model at the Genome Pharmaceuticals Institute. He then returned to the University of Tokyo as an assistant professor. He analyzed the mechanism of lysocin E (2008-2016), finding that lysocin E has a unique mode of action that is distinct from other known antibiotics. Presently, he is an associate professor at the Teikyo University Institute of Medical Mycology. He recently determined that host–microbe interaction enhances the therapeutic efficacy of lysocin E and elucidated the detailed mechanisms involved in this finding, which have been published in Nature Communications.

Department of Food Science and Nutrition, Faculty of the Environment, University of the Aegean, Greece
Efstathios (Stathis) Giaouris has a PhD in Agricultural Sciences from the Agricultural University of Athens (2008) and is currently Associate Professor in Food Microbiology at the Department of Food Science and Nutrition of the University of the Aegean (Lemnos Island, Greece). He has published more than 50 scientific articles in international peer-reviewed journals (h-index = 22, scopus), 8 chapters in international collective volumes, and has announced his work more than 90 times in national, international conferences and workshops. His research focusses on pathogenic bacterial biofilms, especially in terms of the risk of this inherent microbial lifestyle for food hygiene and safety, and their control using novel, cost-efficient and sustainable methods (e.g., phytochemicals, QS inhibitors). Other in parallel research priorities comprise the study of the antimicrobial resistance, the pathogenic microbial physiology under hostile conditions (focused on stress response and virulence induction), the study of the intercellular interactions and their effects on the microbial behavior and physiology, the study and exploitation of beneficial microorganisms for food and health applications. SCOPUS ID: 14032869700; Orcid ID: 0000-0002-6019-5204

Dr. Véronique SINOU is currently a Research Engineer at Aix-Marseille Université with PhD degree in Parasitology from the National Museum of Natural History in Paris. She worked for many years on malaria drug resistance before joining in 2018 the MCT lab to lead the BAC-SCREEN platform which is devoted to high-content screening (HCS) on whole-cell bacteria. Her research aims to identify and develop new bioactive compounds which can fight against resistant microorganisms and the investigation of their mode of action. She focuses on biofilms and the efflux pump systems

School of Food Science and Engineering, South China University of Technology, China
Dr. Xu obtained a B.S. and Ph.D. from South China University of Technology in 2005 and 2011, respectively, and has worked as an Assistant Professor since 2011. Dr. Xu was awarded a Top 100 National Outstanding Doctoral Dissertation in 2014. His major research interests include microbial biofilm, antimicrobial resistance, polymicrobial interaction, and rapid detection. Dr. Xu is the co-founder and co-president of Asia-Pacific Biofilms (previously known as ChinaBiofilms). Dr. Xu has published more than 100 manuscripts as first or correspondence author, with the total IF > 260, citation > 5,000, H-Index as 42, and I-10 Index as 82. Professor, South China University of Technology, Guangzhou, China. Assistant Visiting Scientist, University of Maryland, College Park, United States. Distinguished Visiting Professor, Rajamangala University of Technology Phra Nakhon, Bangkok, Thailand. Adjunct Professor, National Institute of Fundamental Science, Kandy, Sri Lanka.

Strategies to design new antibacterial agents.
Dr. Vila is currently the Head of the Department of Clinical Microbiology of the Hospital Clinic in Barcelona, Full Professor of the School of Medicine, University of Barcelona, and Research Professor in the Institute for Global Health (ISGlobal) of Barcelona, Spain. His present main field of interest is in the investigation of the molecular bases of antimicrobial resistance and the development of new drugs against bacteria and molecular tools for rapid diagnosis of infectious diseases. Dr. Vila was the Programme Director of the Congress of the European Society of Clinical Microbiology (ESCMID) from 2009 to 2014 and he has been a member of the ICAAC Scientific Program Committee (USA) for 4 years. He received an Award of the National Plan against Antibiotic Resistance (PRAN) 2018 for the Micro-combat card game, presented by the Barcelona Global Health Institute Foundation (ISGlobal) in the category of better communication and public awareness initiative on the antibiotic resistance. He is also leading the Initiative of Antimicrobial Resistance in ISGlobal. He has published 476 articles in peer-reviewed journals, 44 book chapters and directed 25 doctoral theses (number of citations, 29.536 and H-index of 84, Google Scholar). He has patented three molecules.

Department of Analytical Chemistry and Food Science, Faculty of Food Science and Technology, University of Vigo, Spain
Jesus Simal-Gandara has been a Full Professor in Food Science at the University of Vigo (Spain) since 1999 and he became the Vice-Chancellor for Internationalization in 2018. He won the 1st Spanish Award of Completion of Pharmacy and the PhD Prize from the Faculty of Pharmacy, University of Santiago de Compostela (Spain). He also was Associate Professor in 1991 at the University of Vigo. Now, he leads a research group of excellence in Northwest Spain. In addition, he was leading CIA3 (Environmental, Agricultural and Food Research Centre) formed by 10 research groups from different fields (botany, plant physiology, soil science and agricultural chemistry, biochemistry and molecular biology, nutrition and food science, biotechnology, food technology, food rheology, chemical engineering, and colloidal chemistry) from 2008 to 2018, and also was the Head of the Department of Analytical Chemistry and Food Science at the University of Vigo between 2013 and 2018. He was also Vice-Chancellor for Internationalization at the University of Vigo in 2018. His research group is specialized in chromatographic separations (GC-MS and LC-MS) and molecular biology and proteomics. They investigate the distribution of agricultural and environmental organic chemical contaminants in the food production chain, and how improving the sensory and functional quality of food, with an eye on the food chain globally, integrating environment, agriculture and food with nutrition and public health issues. His focus today is on the study of persistent organic pollutants (POPs) from the point of view of public health (epidemiology, toxicity of mixtures, metabolites, etc.), and on the study of secondary metabolites in plant foods, exploring the molecular mechanisms that explain their activity, in order to develop new nutraceuticals, functional foods and cosmetics.

School of Chemistry and Molecular Bioscience, University of Wollongong, Wollongong, Australia
Aaron Oakley graduated with a PhD from the University of Melbourne in 1997. He helped to establish a new structural biology laboratory at the University of Western Australia from 1998 to 2003, solving a wide range of structures from mosquito-detoxifying enzymes responsible for pesticide resistance to bacterial dehalogenases for bioremediation applications. He was appointed Fellow at the Research School of Chemistry at the Australian National University in 2004. After a two-year interlude at the Division of Molecular and Health Technologies in the Commonwealth Scientific and Industrial Research Organisation (2008–2009), he was in 2010 awarded an ARC Future Fellowship to establish structural biology at the University of Wollongong. The laboratory moved to the new Molecular Horizons Institute in that university in 2019.
Sessions
S2. Making Old Antibiotics Great Again: How to Reverse Resistance to Antibiotics
S3. New Antimicrobial Approaches, Targets and Mechanisms of Action
S4. Social Challenges and Political Strategies to Prevent Antibiotic Resistance
S5. Antimicrobial Testing and Rapid Diagnostics
S6. Impact of Inter-relationship between Viral and Bacterial Infections
S7. Poster Session
Instructions for Authors
The authors should submit submissions online by logging in ECA2023.sciforum.net and using the “Submit Abstract” function once logged into the system.
- Scholars interested in participating with the conference can submit their abstract (about 200–250 words covering the areas of manuscripts for the proceedings issue) online at this website up to 15 Oct 2023.
- The Conference Committee will conduct a pre-evaluation, based on the submitted abstract, of whether the contribution from the authors of the abstract will be welcome for the 3rd international electronic conference on antibiotics. All authors will be notified by 20 Oct 2023 about the acceptance of their abstract.
- If the abstract is accepted for this conference, the author will be asked to submit a minimum of one of the following: a proceedings paper (3-6 pages), a poster, a slides presentation (in PDF), or a short video presentation (max. 3–5 minutes). This should be ready by the submission deadline of 29 Oct 2023. If you submit a proceedings paper, you need to upload the Word and PDF versions in Original files (zip/word) and Manuscript PDF, respectively. If you choose to submit a poster, a slides presentation (in PDF), or a short video presentation (max. 3–5 minutes), please submit the abstract in Word and PDF format in Original files (zip/word) and Manuscript PDF, respectively. Authors will receive a notification about the acceptance of their papers by 15 Nov 2023
- The manuscripts and presentations will be available on ECA2023.sciforum.net for discussion and rating during the conference from 1–15 Dec 2023.
- All submissions will be reviewed using the powerful text comparison tool iThenticate. This procedure aims to prevent scholarly and professional plagiarism. Submissions will then be peer-reviewed by conference committees based on originality/novelty, quality of presentation, scientific soundness, interest to the readers, overall merit and English level. After the conference, all submissions will be published on sciforum.net, and only the proceeding paper (3-6 pages) will be published in the MDPI Medical Sciences Forum journal (ISSN: 2673-9992)
Note: Publication of proceedings paper is free of charge.
Before publication, Medical Sciences Forum journal will check the plagiarism issue again. Submissions with a lack of novelty will not be published in the journal. - The open access journal Antibiotics published a dedicated conference Special Issue: Rise of Antibiotic Resistance: Mechanisms Involved and Solutions to Tackle It. Conference participants are encouraged to submit a full paper to the dedicated Special Issue and will receive a 20% discount on the Article Processing Charges (APC).
Submitted manuscripts should not have been published previously nor be under consideration for publication elsewhere. Manuscripts for the proceedings issue must have the following organization:
- Title
- Full author names
- Affiliations (including full postal address) and authors' e-mail addresses
- Abstract (200–250 words)
- Keywords
- Introduction
- Methods
- Results and Discussion
- Conclusions
- (Acknowledgements)
- References
Authors are encouraged to prepare a presentation in PowerPoint or similar software to be displayed online along with the Manuscript. Slides, if available, will be displayed directly on the website using Sciforum.net's proprietary slides viewer. Slides can be prepared in exactly the same way as for any traditional conference where research results can be presented. Slides should be converted to PDF format before submission so that our process can easily and automatically convert them for online display.
Authors are also encouraged to submit video presentations. This is a unique way of presenting your paper and discussing it with peers from all over the world. The video should be no longer than 3-5 minutes and prepared with one of the following formats: .mp4 / .webm / .ogg (max size: 250Mb). It should be submitted with the full manuscript before 29 Oct 2023.
2)The minimum size for images is 148 mm × 210 mm (horizontal × vertical) at 300 dpi.
3)The content of the poster should be a comprehensive presentation of your accepted submission.
4) There should be no copyright issues with any elements in the poster.
It is the author’s responsibility to identify and declare any personal circumstances or interests that may be perceived as inappropriately influencing the representation or interpretation of clinical research. If there is no conflict, please state here “The authors declare no conflict of interest”. This should be conveyed in a separate “Conflict of Interest” statement preceding the “Acknowledgments” and “References” sections at the end of the manuscript. Financial support for the study must be fully disclosed under the “Acknowledgments” section.
MDPI, the publisher of the Sciforum.net platform, is an open access publisher. We believe that authors should retain the copyright to their scholarly works. Hence, by submitting a Communication paper to this conference, you retain the copyright of your paper. Still, you grant MDPI the non-exclusive right to publish this paper online on the Sciforum.net platform. This means you can easily submit your paper to any scientific journal at a later stage and transfer the copyright to its publisher (if required by that publisher).
Live Sessions Recordings

Event Awards

The Awards
Number of Awards Available: 1
As a sponsor, Antibiotics would like to award the best paper as elected by the conference committee. The award will consist of 500 Swiss Francs. We look forward to posting your contributions.
Criteria for Evaluation of Best Paper Award:
· Full paper must be submitted to ECA 2023;
· Originality/novelty of the paper;
· Significance of content;
· Scientific soundness;
· Interest to the readers;
· English language and style.
Evaluation
· Each Evaluation Committee member will give an assessment for each applicant in terms of the criteria outlined above;
· The total score for each presentation will be ranked from highest to lowest;
· If two or more authors receive the same score, further evaluation will be carried out;
· All decisions made by the Evaluation Committee are final.
Number of Awards Available: 1
As a sponsor, Antibiotics would like to grant an award (500 Swiss Francs) for the best poster at the conference. This prize will be determined by a jury and awarded to the best-designed poster presented at the conference.
Criteria for Evaluation of Best Poster Award:
· Title (with authors and affiliations)
· Introduction/Objectives/Aims
· Methods
· Results
· Conclusion
· References
· Acknowledgements
· Contact information
Evaluation
For consideration for this award, posters should be accompanied by a three-minute video presentation. During the conference, the chair will be invited to judge the quality of the video presentations and posters. Presentations will be judged on how well they are able to summarize the content of the work and make the observer interested in viewing the poster. Posters will be judged on clarity quality of appearance.
Conference Secretariat
Ms. Jocelyn Yang
Ms. Kelsey Li
Mr. Russell Wang
Email: eca2023@mdpi.com
For inquiries regarding submissions and sponsorship opportunities, please feel free to contact us.
S1. Epidemiology, Prevalence and Mechanisms of Microbial Resistance
Session Chair
Dr. Nicholas Dixon, School of Chemistry and Molecular Bioscience, University of Wollongong, Australia
Show all published submissions (11) Hide published submissions (11)
Submissions
List of Papers (11) Toggle list
S2. Making Old Antibiotics Great Again: How to Reverse Resistance to Antibiotics
Session Chair
Dr. Anthony Coleman, Laboratoire des Multimatériaux et Interfaces, France
Show all published submissions (5) Hide published submissions (5)
Submissions
List of Papers (5) Toggle list
S3. New Antimicrobial Approaches, Targets and Mechanisms of Action
Session Chair
Dr. Jordi Vila, Department of Clinical Microbiology, Hospital Clinic, Spain
Show all published submissions (11) Hide published submissions (11)
Submissions
List of Papers (11) Toggle list
S4. Social Challenges and Political Strategies to Prevent Antibiotic Resistance
Session Chair
Dr. Efstathios Giaouris, Department of Food Science and Nutrition, Faculty of the Environment, University of the Aegean, Greece
Show all published submissions (5) Hide published submissions (5)
Submissions
List of Papers (5) Toggle list
S5. Antimicrobial Testing and Rapid Diagnostics
Session Chair
Dr. Zhenbo Xu, School of Food Science and Engineering, South China University of Technology, China
Show all published submissions (2) Hide published submissions (2)
Submissions
List of Papers (2) Toggle list
S6. Impact of Inter-relationship between Viral and Bacterial Infections
Session Chair
Dr. Jean Marc Sabatier, Institute of Neuro Physiopathology, France
Show all published submissions (1) Hide published submissions (1)
Submissions
List of Papers (1) Toggle list
S7. Poster Session
Show all published submissions (42) Hide published submissions (42)
Submissions
List of Papers (42) Toggle list